Making Multi Layer Tablets
Find innovative production technology for making multi-layer tablets and connect directly with world-leading specialists
Multilayer tablets are a popular fixed-dose combination (FDC) offering a range of release profiles for effective drug delivery. Multilayer tablet technology solves challenges to API consistency, allowing manufacturers to develop formulations from incompatible ingredients while optimizing the release profile for immediate release and controlled release.
Select your multi-layer tablets process
Tell us about your production challenge
Use accurate weight-dosing systems and excipients to avoid delamination
Tablet sequencing is vital to achieving the correct lamination in the inner layer of the capping. Multi-layer technology relies on the accuracy of weight-dosing systems to counter delamination.
An inadequate ratio of the layer sizes presents an additional challenge to layering. The target ratio is 1:1, but API incompatibility and separate formulation mixtures impact the sizing of layers. Compressibility, flowability, and particle size distribution are crucial to controlling the weight and size of different layers.

Reduce cross-contamination with gluing pills technology to ensure adhesion between layers
Gluing pills technology (GPT) uses a binding agent to attach both layers after they’ve been individually compacted. The binding process safeguards against cross-contamination and ensures both tablet layers do not interact with one another.
Cross-contamination is a significant risk in formulation design when active pharmaceutical ingredients are exposed to other materials.

Superdisintegrants facilitate repeat-action of multilayer tablets
A tablet-within-tablet design makes multilayer formulations a good solution for repeat-action products. But their pharmacological effectiveness depends on the correct disintegration ability of the outer tablet.
Traditionally, starches and clays have been used as disintegrants by attracting water into the tablet. However, these may occupy up to a fifth of the weight of the final tablet. Specially developed disintegrants like Crospividone, Croscarmellose Sodium and Sodium Starch Glycote offer higher effectiveness at a lower concentration. These three make up the main categories of superdisintegrants.
Multilayer tablet technology is ideal for polypharmacy in single dose form
Polypharmacy is a growing area of interest in medication. It finds an effective solution in multilayer tablets by administering a minimum of 5 medicines at regular intervals.
Formulas designed for this purpose may include high-potency active pharmaceutical ingredients (HPAPI) with smaller but more powerful concentrations of materials. Multilayer tablet technology combines these complex formulations to manufacture more efficacious pharmaceutical products.
Processing steps involved in multi-layer tablets making
Which multi-layer tablets technology do you need?

Laboratory film coating system for development and clinical batches
Achieve precision in tablet coating with a versatil...

Tablet hardness tester
Ensure consistent tablet quality by precisely measuring hardness, thickness, diameter, and weight with...

Tablet hardness and combination tester
Ensure consistent tablet hardness and precision across multiple parameters with this...

Tablet press for multi-layer and dry-coated tablets
Optimize your tablet production with high-speed multi-layer pressing ...

Single rotary tablet press
Streamline your tablet production with enhanced efficiency and precision, ensuring consistent tab...

Flat-forming blister packager for small production batches
Ideal for pharmaceutical and nutraceutical firms seeking effi...

High-output blister packaging system
Maximize blister packaging efficiency with continuous-motion capabilities, ensuring su...

Intermittent motion cartoning machine for blister packs
Optimize your packaging line with a space-efficient cartoning mac...

Semi-automatic de-blistering solution for Pvc and alu blister packs
Streamline your packaging operations by swiftly rec...

Automated sample preparation workstation for content uniformity testing
Streamline your laboratory workflows with autom...

Agitator bead mill for API
To achieve precisely defined API properties and safe and reproducible production, rigorous implem...

Continuous wet granulator and dryer for R&D
Pharmaceutical laboratories need compact equipment to handle and produce small...

Dry granulator for pharmaceutical powders
In pharmaceutical industries, where large-volume production is the norm, compact...

Energy-efficient film coating machine for pharmaceutical tablets
Tablet coating involves applying a thin layer of coatin...

Pharmaceutical tablet coating machine
Tablet coating requires spraying, mixing, and drying procedures that must be carried ...

Single-use containment valves
From GMP and HSE perspective material transfer is critical for chemical and pharmaceutical for...
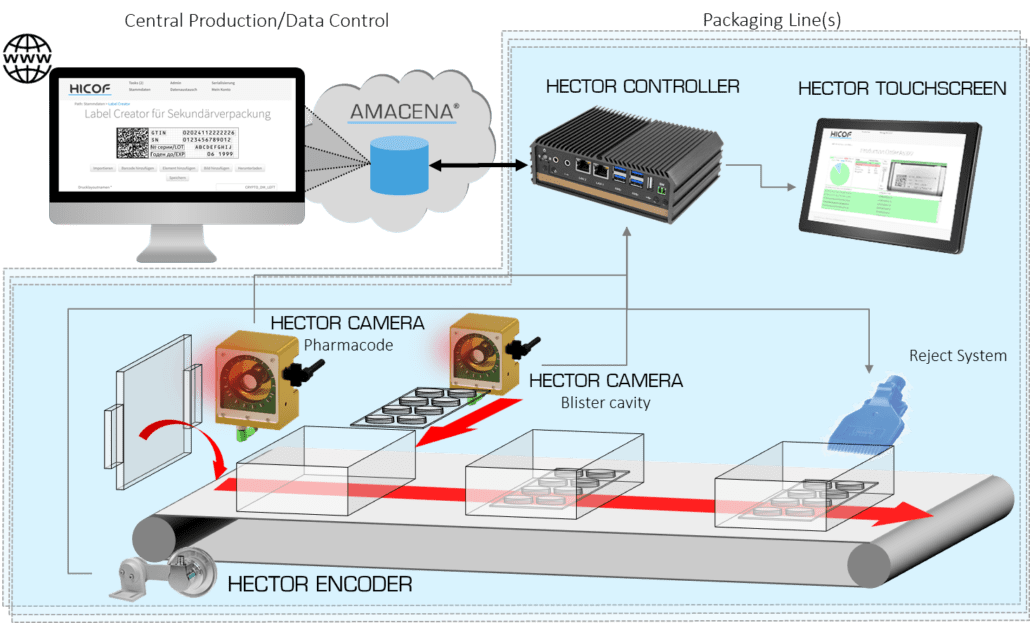
Serialization and aggregation inspection system
Including serialization and aggregation inspection systems to packaging li...

Serialization and aggregation system
Manufacturers from the pharmaceutical industry require a variety of cartons, cases and...

High speed bottle filler with inspection
Medicines in tablet, capsule, or soft gel forms undergo various processes before r...

Lifter and De-duster for Tablets
The use of several different tools for lifting, de-dusting, and polishing in the pharmaceut...

High Speed Visual Inspection System for Tablets
In order to ensure quality and standard specifications of tablets, capsule...

Contained Visual Inspection System
In the medicine industry, it is important to ensure there are no defects in tablets or c...

Industrial scale tablet coater
During the coating process, tablets pass many stages that can make them wet and sticky or cra...

Tablet counting machine
Production of pharmaceutical solid dosage forms usually require complete accuracy, flexibility, and c...

High Speed Double-Sided Tablet Press
To achieve as much as double the output of single-sided tablet press, the upper and lo...

Laboratory Tablet Press
Pharmaceutical research and development projects may require small samples of high quality tablets fo...

Dust-tight metal detector for tablets
For FDA-compliant production of tablets, the elimination of airborne dust containing ...

Benchtop tablet press
For lab work or small-scale production, a purpose-designed tablet press that meets your exact requireme...

Semi-automatic case packer
Pharmaceutical products require complete traceability throughout the production cycle. For smalle...

Stand-alone aggregation station
The aggregation of pharmaceutical products for tracking purposes is already compulsory in ma...

Serialization coding and labeling equipment
In the pharmaceutical industry, product serialization is the cornerstone of al...

Single rotary tablet press
Creating batches of consistent high quality pharmaceutical grade tablets from your powdered ingre...

Small feeder with flexible wall hopper
In many laboratory applications and production processes, smaller quantities of powd...

Automatic tablet coater for lab scale
The ability to coat tablets in an even and controlled way is an important stage in th...

Tablet auto coater for lab scale
Many pharmaceutical doses in tablet form require a coating before they are ready for use. T...

Single and double layer tablet press
High performance tablet presses that are capable of producing both single and double-l...

Granulation line of mixer and fluid bed dryer
A complete granulation line allows for the mixing and granulation of pharmac...

Rotary tablet press
For pharmaceutical tablet producers looking to reduce waste, costs and increase production volume, a hig...

Fluid bed dryer for production scale
Fluidized bed drying (FBD) is a common process in the pharmaceutical industry for dryi...

Entry-level high-shear mixer for drug formulation
For pilot and lab-scale pharmaceutical formulations, a high-shear granu...

R&D electronic counter for capsules and tablets
This machine has been designed to offer an ever-precise and reliable count...

Premium vacuum conveyor
When you have a need to tailor make your conveyor and still have the high requirement on hygiene, e.g...

High containment split butterfly valve
Powder containment is the cornerstone of chemical and pharmaceutical manufacturing. ...

Sorption dehumidifier with air-chilled condenser
It is not always possible to duct out the wet air from a dehumidifier ser...

Small portable dehumidifier
Water damaged areas require dehumidification to limit the damage and prepare the area for restor...

Sorption dehumidifier for large temperature differences
Small, humid areas can be difficult to dehumidify in humid ambien...

Sorption dehumidifier for deep drying
Humid ambient conditions such as those in tropical climates make dehumidification mor...

Dehumidifier for difficult wet airflows
Sealed rooms can be challenging to dehumidify with ordinary dehumidifiers. The wet ...

Sorption dehumidifier for overpressured rooms
Small humid spaces, used for processing or storage, often need a simple dehu...

Heat recycling sorption dehumidifier
Facilities that handle large amounts of water can become very humid. High humidity may...

Industrial desiccant dehumidifier
Dehumidification of medium sized manufacturing and processing facilities can be a dauntin...

Large scale desiccant dehumidifier
Moisture sensitive processing and storage facilities need low humidity environments to s...

Customizable dehumidifier
Dehumidification needs can vary significantly from one project to another. Dehumidifiers with a fi...

Checkweigher
A check weigher that is equipped with high speed/high accuracy force balance load cell. It meets stringent accura...

Entry-level X-ray inspection system
Without compromising on performance, if you need an entry level x-ray inspection system...





















